Solved Problem on Heat
advertisement
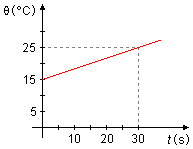
A body of mass 200 g is heated for 30 seconds by an energy source that provides a power of 210 W
at a constant rate. Given the graph of temperature as a function of time, determine the heat capacity
of the body, knowing that 1 cal = 4.2 J.
Problem data:
- Mass of the body: m = 200 g;
- Power of the heat source: \( \mathscr{P} \) = 210 W.
First, we must convert the power unit given in watts (joules per second) to calories per second (cal/s), in this problem, it is more convenient not to use the International System of Units (SI)
\[
\begin{gather}
\mathscr P=210\;\mathrm W=210\;\mathrm{\frac{J}{s}}=210\;\mathrm{\cancel J}\;\frac{1\;\mathrm{cal}}{4,2\;\mathrm{\cancel J}}\times\frac{1}{\mathrm s}=50\;\mathrm{\frac{cal}{s}}
\end{gather}
\]
The heat capacity is given by
\[
\begin{gather}
\bbox[#99CCFF,10px]
{C=mc} \tag{I}
\end{gather}
\]
The quantity of heat received (or lost) is given by
\[
\begin{gather}
\bbox[#99CCFF,10px]
{Q=mc\Delta \theta } \tag{II}
\end{gather}
\]
where θ was used for temperature in place of t was used for the time in the problem.
Substituting the expression (I) into (II)
\[
\begin{gather}
Q=C \Delta\theta \\[5pt]
C=\frac{Q}{\Delta\theta} \tag{III}
\end{gather}
\]
During the heating time, by the graph, we see that the temperature varied from
\[
\begin{gather}
\Delta\theta=\theta_f-\theta_i \\[5pt]
\Delta\theta=(25-15)\;\mathrm{°C} \\[5pt]
\Delta\theta=10\;\mathrm{°C} \tag{IV}
\end{gather}
\]
The heat received by the body is obtained from the power of the energy source. The source provides 50
calories in 1 second, so the 30 second heating will provide a quantity Q of heat, using
Cross-multiplication
\[
\begin{gather}
\frac{50\;\mathrm{cal}}{1\;\mathrm s}=\frac{Q}{30\;\mathrm s}
\end{gather}
\]
performing the cross multiplication
\[
\begin{gather}
(1\;\mathrm s)\times(Q)=(30\;\mathrm s)\times(50\;\mathrm{cal}) \\[5pt]
Q=\frac{(30\;\mathrm{\cancel s})\times(50\;\mathrm{cal})}{1\;\mathrm{\cancel s}} \\[5pt]
Q=1500\;\mathrm{cal} \tag{V}
\end{gather}
\]
substituting the values (IV) and (V) into equation (III), the heat capacity will be
\[
\begin{gather}
C=\frac{150\cancel 0\;\mathrm{cal}}{1\cancel 0\;\mathrm{°C}}
\end{gather}
\]
\[
\begin{gather}
\bbox[#FFCCCC,10px]
{C=150\;\mathrm{cal/°C}}
\end{gather}
\]
advertisement

Fisicaexe - Physics Solved Problems by Elcio Brandani Mondadori is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License .